 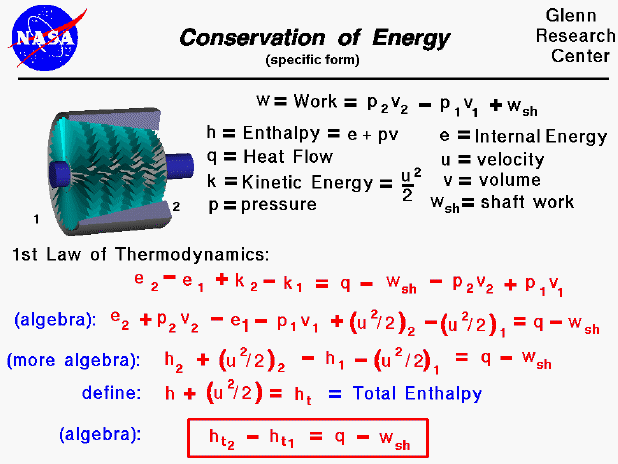
The increase of disorder provides most of the free energy. ΔH The free energy comes mostly from a flow of thermal energy. ΔH > T * ΔS then the reaction is enthalpy-driven.The direction of a free energy change can be either enthalpy- or entropy-driven. ΔG0 - a nonspontaneous process - additional energy must put in for the reaction to happen (a round boulder being pushed up a hill).We can also define it with regards to the change in free energy: The Gibbs free energy equation is:Įarlier, we talked about spontaneity of a process and how it is associated with entropy. 
It's a function of both enthalpy and entropy, and is used to predict the spontaneity of a processes. What is Gibbs free energy? It's the energy in a system available to do work on its surroundings at constant pressure and temperature. Every system tends toward stability, and, for an irreversible process, maximum stability is reached it when the system's energy is most disordered. 
The entropy of a system is strictly connected to the systems energy. As stated by a physicist Rudolf Clausius: "The entropy of the universe tends to a maximum." You intuitively know that the opposite process is not possible - the milk won't separate from coffee by itself.Īny spontaneous process increases the disorder of the universe. You observed that the milk quickly mixes with the coffee. Let's say you've made yourself a hot cup of coffee. It might sound complicated, but you'll understand it easily with an everyday example. It doesn't have to be a fast - it can even be still occurring when the heat death of the universe occurs - but if it would proceed without the addition of any outside energy, it's spontaneous. It's one of the main determinants of the spontaneity of a reaction.Ī spontaneous process is one that doesn't require an outside source of energy to proceed. But why measure disorder, and is it even possible? Physically, we can't measure entropy, but we can calculate it. *I thank Stephen Selipsky for bringing Page's results to my attention and for his patient explanations.Entropy is the measure of that disorder. As the universe expands and cools, however, eventually the black hole may begin to lose mass-energy through Hawking radiation. Indeed, any black hole with a mass greater than about 0.75% of the Earth's mass is colder than the cosmic background, and thus its mass increases for now. So rather than shrinking, it would continue to grow. Therefore, whatever little energy it radiates, it actually receives more in the form of heat from the cosmos. But that does not take into account the fact that such a black hole is colder than the cosmic microwave background radiation bathing it. The lifetime of a $1~M_\odot$ black hole, therefore, is calculated as nearly 57 orders of magnitude longer than the present age of the universe. The latest version also correctly accounts for the black hole's effective area, light scattering, and the resulting change in its evaporation lifetime. The drop-down menus select the units of measure to be used for their corresponding input field.Īn added feature is the calculation of the "peak photon" wavelength, corresponding frequency, and photon energy, representing the peak of the blackbody radiation curve per unit logarithm (of wavelength or frequency) that corresponds to the black hole temperature. I kept the unit, but decided to use instead the much more useful value of one solar mass as the initial mass.Īs in Wisniewski's version, specifying any quantity causes the others to be recalculated accordingly (see source). Wisniewski's original code included a fictitious unit of mass, the "standard industrial neuble", equivalent to a billion metric tons, from Will McCarthy's novel The Collapsium. The original idea belongs to Jim Wisniewski, whose page from 2006 ( link) appears to be no longer available available again, but since it is not archived by the Wayback Machine, I think my functional clone is still useful. This page contains a JavaScript calculator of Hawking radiation and other parameters of a Schwarzschild black hole. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
